Expert Q&A: Dr. Richard Ostfeld
By Amy Nelson
Richard Ostfeld is Distinguished Senior Scientist at the Cary Institute of Ecosystem Studies. He has published more than 200 peer-reviewed articles related to disease ecology, and he is the author of the book Lyme Disease: The Ecology of a Complex System (2011, Oxford University Press). Ostfeld’s research explores the ecology of infectious diseases; linkages between community ecology and epidemiology; behavioral, population, and community ecology of mammals; animal population dynamics; and landscape epidemiology. He has given several USA Congressional Briefings on environmental change and emerging infectious diseases. His work sheds light on the power of biodiversity to protect human health by reducing the transmission of zoonotic diseases, and we were thrilled to have the chance to speak with him to learn more.
What is disease ecology, and how did you become interested in it?
Disease ecology is a new subdiscipline within the scientific discipline of ecology. It focuses specifically on the causes and consequences of disease patterns in organisms (including humans.) Ecology is the scientific discipline that focuses on interactions between organisms in nature. If you think about diseases—especially infectious diseases—you realize that they are a quintessential case of interactions between species in nature. There are pathogens that are distinct organisms, and hosts that are other distinct organisms, and the interaction between them is what causes the disease.
Infectious disease has tended to be the purview of the biomedical community. Indeed, biomedical approaches are extremely important, but in the past 20 to 25 years, ecologists have realized that we have a very strong role to play in understanding where diseases come from, how they affect populations of animals (including us), plants, and microbes. This is a very active, rapidly growing area in the scientific discipline of ecology.

Many Leaf Litter readers in North America do a lot of field work and are quite familiar with ticks. Unfortunately, many of them have also had personal experiences with Lyme disease. Can you briefly describe the ecology of Lyme disease?
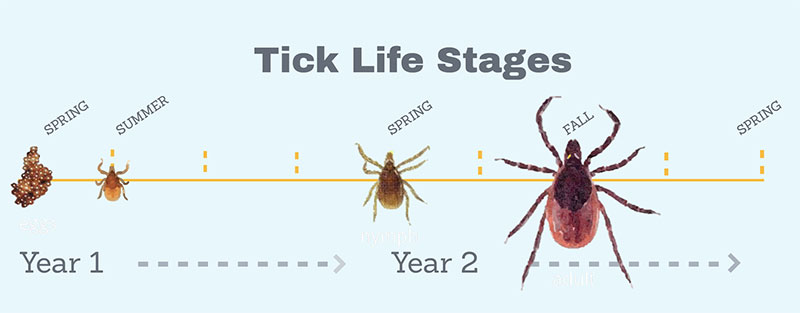
Lyme disease is acquired by the bite of an infected tick (Ixodes scapularis). It’s usually the black legged tick in most of North America. There are related species of tick in Europe, Asia, and the west coast of the US, that transmit the agent that causes Lyme, which is a bacterium (Borrelia burgdorferi).

Many people don’t realize that baby ticks hatch out of their eggs free of infection; mother ticks infected with the Lyme disease bacterium don’t pass it on to their offspring. For a tick to become infected, and therefore dangerous to us, it has to acquire the bacteria from an animal – often from small rodents, especially white-footed mice (Peromyscus leucopus) but also from eastern chipmunks (Tamias striatus).

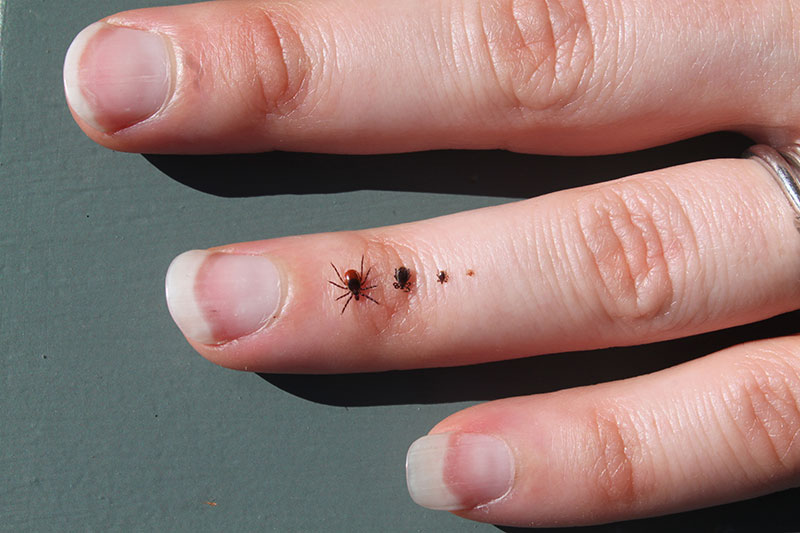
The baby ticks bite the mice, and the mice often transmit the infection to those larval stage ticks. Those larvae then appear the next year as nymphs. In the nymphal stage, ticks are tiny like the larva, but a little bigger, about the size of a poppy seed. At this stage, ticks typically come out in May or June and are able to infect people and wildlife. What we try to understand is what determines how many infected nymph-stage ticks are out in the environment. These numbers vary from year to year and place to place. We study things like the role of fluctuating white-footed mice, the role of predators on those mice, the role of climate change, and the role of different types of management of the disease as well.
What have you learned about the role of habitat destruction and the spread of Lyme disease?
Habitat destruction is a very important part of the Lyme disease phenomenon, and it ultimately plays out through the impact of white-footed mice. As I’ve said, mice are the main source of infection for ticks. Mice are also very hospitable hosts for those baby ticks; they do not groom them off once attached. Ticks trying to bite a mouse tend to survive, as opposed to ticks trying to bite some other mammal or bird, which tend to be better at grooming them off and killing them. More mice lead to more nymph-stage ticks. Because mice are the main source of infection, more mice also means higher, increasing infection prevalence among nymph-stage ticks. The mice are kind of a double whammy.
Here’s where habitat fragmentation comes in. It turns out that when we fragment or degrade the forest, we tend to reduce diversity of animals in the landscape. And the species we tend to lose first are the larger bodied, more predatory species that require large, extensive areas of habitat to maintain a viable population. Those species are often capable of regulating the size of the mouse population, by eating them, or relegating them to very small, specialized parts of the environment, or, in some cases, competing with them. When we lose the species that can control the mice, the mice thrive. And, indeed, mice do really well in parts of our landscape that we’ve fragmented or degraded. They’re the last species to go, and therefore serve as the most available blood meals for hungry ticks. Feeding ticks become infected, increasing our risk of exposure. Where the habitat is not fragmented, many of the ticks feed on other hosts but not mice, and we see fewer infected ticks as a result.
Is what you just described the “dilution effect?”
The dilution effect is a term that Felicia Keesing and I coined back in the late nineties to refer to the fact that high wildlife diversity dilutes the impact of white-footed mice and reduces human risk of exposure to Lyme and other tick borne diseases. That’s for the mechanisms that I just described. In some cases, the loss of diversity means the loss of species that do a good job of killing the ticks that try to feed on them. And so we’re losing the species that actually do us a favor by killing ticks out in the environment. Ticks that feed successfully on this diverse wildlife—like opossums, skunks, foxes, and bobcat—do not get infected at a very high rate, so even if they survive, they don’t increase disease risk. The diverse wildlife in our better protected landscapes help us. They protect us by reducing tick numbers and reducing tick infections. They’re diluting the role that white-footed mice play.

To what extent is the dilution effect factored into policies, forest management plans, or things like the management of hunting seasons?
I don’t think it has been incorporated to the degree that it could be. There are international efforts like the Intergovernmental Platform for Biodiversity and Ecosystem Services (PBES) that refer to the dilution effect but it does not tend to make it into specific recommendations. It is challenging to apply the dilution effect to specific local or regional regulations for planning or zoning. They don’t necessarily match up.

We have found that forest fragments of a couple of acres or smaller are the riskiest possible places with the highest densities of infected ticks. I have had planning boards and zoning boards discuss this with me. But they interpret this as meaning that they should have larger zoning lots—say five-acres, rather than one acre. That doesn’t necessarily address the problem, because every five-acre parcel could have a one-acre woodlot on it, and that would pose a devastatingly high risk. Knowing that small forest fragments and fragmented parts of the landscape are very dangerous does not necessarily translate directly into zoning parcel size. Applying our scientific knowledge adequately to use in regulations, planning, and policy is a tricky business, and very difficult to do.
I’ve been very busy working along with Felicia Keesing on the Tick Project, where we are trying to engage communities to actively reduce Lyme disease risk using environmentally safe methods. [Note: The Tick Project is a five-year study taking place in Dutchess County, NY to determine whether neighborhood-based prevention can reduce human cases of Lyme and other tick-borne diseases.]

What role might ecological restoration play in restoring habitat for the purpose of restoring the dilution effect?
That is a great question, and it’s under-explored. I get asked a lot about re-introducing certain species, such as opossums, that we know can reduce the abundance of infected ticks. Unfortunately, managing our landscapes for particular wildlife species is difficult. We can’t just put out opossum nest boxes and think that we’re suddenly going to boost those populations. The piecemeal approach of trying to manage for specific species is impractical and might not be very effective restoration.
But corridors that would connect more extensive forests—forests that maintain high diversity—would allow that diversity to persist and those species to spread into nearby habitat areas, and that could have a beneficial effect. It would need to be tested, though, and there is very little, if any, funding for that kind of work. Looking experimentally at different types of landscape management or restoration programs to ask how they affect risk of tick-borne disease would be very valuable. It would also be very expensive, and it falls between the cracks in our federal funding system, landing between ecology and health sciences. That is a problem of the siloed way we do science in this country.

What other zoonotic diseases are subject to the dilution effect, and what else did you learn about the spread of Lyme disease that can be helpful in understanding the outbreak and spread of other zoonotic pathogens?
Quite a few other zoonotic diseases seem to be subject to the dilution effect. With West Nile virus, which is transmitted by mosquitoes (mostly Culex), we know that the higher the diversity of birds, the lower the West Nile infection prevalence in mosquito populations, which can lower the incidence rate in human populations in those areas.
The dilution effect occurs in Hantaviruses, which are transmitted typically by rodents. Hantaviruses are a problem all over the world, and they can be very debilitating and deadly. We know that higher small mammal diversity lowers the probability that humans get sick with Hantaviruses. I would mention one other disease, which is called Chagas disease, which is a protozoal disease transmitted by an insect called a kissing bug (Triatominae). And this is very prevalent. Hundreds of millions of people are at risk, especially in Latin America. With Chagas disease, we find that when diversity is lost, the small rodent reservoir hosts that support the parasite, become very abundant. They bourgeon, and disease risk goes up. So a similar pattern is found for both vector borne diseases, like West Nile virus, but, also, non-vector-borne diseases like Hantavirus.
It is clear that our encroachment into wildlife habitat increases the likelihood that people would come into contact with zoonotic pathogens. What other factors related to habitat destruction may come into play in the spread of zoonotic diseases? What do we know about the physiological and behavior changes that animals experience in response to alteration of their habitat by humans, and do those changes make them more vulnerable to pathogens?
Habitat destruction and disturbance can have multiple impacts on infectious disease. One is this dilution effect, wherein the abundance of infected vectors or the abundance of infected hosts increases. If you enter those areas, you are more likely to get exposed.
Habitat disturbance also makes human entry more likely. If we’re building logging roads or putting new soybean fields or cattle pastures where we’ve replaced native forest, human incursions into those newly created danger zones will be more likely.

Sometimes when we destroy or degrade natural habitat, we disturb the animal populations that still persist. This has been seen in some viral diseases transmitted by bats, such as Nipah virus and Hendra virus. Bats are naturally infected with these viruses. They don’t seem to get sick from them. But when we encroach on that habitat, we force them to fly farther to reach food sources and roosting trees. We also disrupt their daily migrations, and there is evidence that their rates of stress go up. We know that stress often suppresses immunity, and in the case of bats, stressed animals tend to shed more virus in their urine, feces, and saliva. It is probably widespread, although it has only been studied in a few specific cases, that stressing native animal populations, even when they are able to persist, can cause a reduction in their ability to ward off infection, which might cause them to be better spreaders. This is bad news for both the wildlife population itself, as well as potential zoonotic transmission to us.
The COVID-19 pandemic seems to have raised awareness of the connection between habitat destruction and human health. How might an increased awareness impact conservation?
I wish I could gaze into a crystal ball and tell you that. I think you’re right that the COVID-19 pandemic has highlighted the role that habitat destruction and degradation plays in changing patterns of infectious disease outbreaks and transmission. There is also a lot of confusion about this, even among scientists and policy makers. There is a sort of mythology that the most biodiverse places on the planet are the riskiest, the idea being that the more species there are, the more dangers there might be. There is a fear of the unknown. There are scientists who fall prey to this fallacy that in the most biodiverse places on the planet, there are more pathogens numerically and therefore risk is higher.
That is not actually the case. There may be more raw numbers of pathogens in biodiverse places, but the vast majority of those pathogens have essentially zero zoonotic potential. They are not things we need to worry about. They circulate in their hosts, and they do not cause problems in those hosts or in people.
It’s actually a restricted set of pathogens that are most dangerous. It tends to be the pathogens that reside in rodents (not just any rodents, but the species that tend to thrive when biodiversity is lost) and secondarily, in bats. It tends to be in species like white-footed mice or other ‘weedy’ species that react in a positive way to habitat destruction and biodiversity loss. Not every host is equally likely to be a risk to us, but the ones that thrive when we reduce diversity tend to be the ones that are filled with pathogens that can make us sick. I’m fond of saying that the next pandemic pathogen is vastly more likely to come from a rat than from a rhino.

There is plenty of empirical support for this. Biodiversity is not a threat, when it comes to infectious disease, as much as it is a buffer against transmission of the rather small subset of pathogens that come from the species that thrive under that reduced biodiversity. We need to distinguish between the mythology and the scientifically supported relationship between diversity and disease. Diversity largely serves a protective role.
What is the greatest need for research in this area?
We need to start applying our understanding of these general principles to specific situations. I would like to see work done incorporating human health impact assessments with environmental impact assessments. This should be done when there is a development project proposed by an international or domestic entity, such as putting in a dam, or increasing cattle ranching or soybean farming—something that would impact natural ecosystems in an important way. We have a process. I think it’s a flawed process, but at least we have one for assessing the environmental impacts of a project, such as how will it influence erosion, water filtration, air quality, and things like that. We need to incorporate impacts on infectious disease risk into this assessment process. Funding could allow environmental health impact assessments to be done in a scientifically rigorous and expansive way.

If a development project is found to pose a risk to human health via infectious disease risk, alongside those other environmental impacts, then the project should be redesigned or stopped. For particular economic development purposes, there are cons to those, and we need to examine more rigorously what those costs are, how far into the future they will extend, and where they will be experienced. Will all the gains of that new development project accrue to people in faraway places who already benefit from huge economic gains? Will all the costs be borne by the local people who will gain none of the benefits? Will it affect one generation, or five? These are questions that can be addressed, and it takes funding to ask them in a rigorous way.
How can practitioners of ecological restoration, conservation, and sustainable design who want to bring this particular public health lens into their work find specialists in this area?
That’s a tough one. Currently, there is no trail that is clearly marked that would allow for people to do that. I am not aware of a good clearinghouse that connects end users to the knowledge producers. There is a real gulf in our communication: the pipeline of science and the use of science. Some of the cooperative extension types of entities are particularly good at that. Many universities have departments with faculty who are practicing scientists and very knowledgeable, but they are also overwhelmingly busy. However, finding individual scientists through searches of publications on the topic can be effective.
I do not know of examples. There are, however, plenty of examples where we determined after the fact that there were horrible public health outcomes of certain development projects. The construction of dams in West Africa and in parts of Latin America, the deforestation of parts of the Amazon Basin: these have resulted in massive outbreaks of schistosomiasis, a snail-transmitted, parasitic disease that largely strikes children, and of Malaria. These are two of the biggest infectious disease problems on the planet right now, and we know there is a very strong link between development projects and the exacerbation of disease in the local communities.
I am not aware of the extent to which that knowledge has been used to either mitigate the environmental causes of elevated risk, or to redesign the next project, or adjust the current project to reduce disease transmission. It seems like a critically important way to use our scientific knowledge for humanity, but I’m not sure it happens. You should keep asking that question. It’s a really important one, and I wish I had a better answer for you.
What do we know about the impacts of climate change on the spread of zoonotic pathogens?
We know that because of climate change, there are species involved in disease transmission that are extending their geographic ranges to higher latitudes. This is true of several species of mosquitoes that are important in disease transmission. I’m referring here to Aedes mosquitoes that transmit dengue fever, chikungunya, yellow fever and Zika viruses, as well as Anopheles mosquitoes that transmit malaria parasites.
Ticks are on the move as well, in part because of climate warming. These cold-blooded species can be limited by the range of conditions that allow them to live out their life cycle, and we are lengthening the season. For ticks, a longer season means more opportunities to find a host, which is essential for survival and population growth. We are increasing the mean temperature and changing patterns of precipitation. More flooding events, more extreme rainfall followed by drought – these patterns can boost certain types of vector populations, especially mosquitoes.
We have diseases moving into new areas where climatic conditions were previously unsuitable. That is a major concern. And it’s not just diseases of humans. We know that avian malaria is moving upslope in the Hawaiian Islands into higher elevations where it simply couldn’t occur before because the mosquitoes could not exist there. But with climate change they can, and the native birds have never experienced this set of malaria parasites, so they are immunologically naive and highly vulnerable. We’re seeing their demise as a result of climate change-induced increases in mosquito populations. We will continue to see more movement of diseases into these vulnerable, immunologically naive host populations, where they will affect both people and wildlife. It’s a daunting prospect.
Are there any alarming human-to-animal diseases that are occurring because of human encroachment into habitat? If so, what impacts might that have on biodiversity?
There are cases of human diseases being transmitted into gorilla and chimpanzee populations. There is concern right now about transmission of the novel coronavirus into primate populations, especially those that are being studied by human researchers or those living in close proximity to people. We know that the Syrian (golden) hamster (Mesocricetus auratus) is vulnerable to infection by this novel coronavirus. That is exciting for scientific researchers, because this is an easily reared creature that is used in a lot of laboratory biomedical studies. But what that suggests is that, potentially, there are other species of rodent that could be vulnerable to infection. We don’t know much about it yet because this is such a novel virus, but it’s something we need to take seriously.

We do need to be careful about the reverse direction of transmission from humans to animals in known situations, as well as for novel pathogens as we discover them.
Do you have any words of encouragement to offer our readers?
I find it personally encouraging that there are co-benefits out there. There are win-win situations; not everything in life is a tradeoff. So here we have a situation where what’s good for the environment—protecting biodiversity, protecting intact habitats—is also good for our health. That is an optimistic message. We can achieve multiple goals that are important to us in the realms of nature, conservation, preservation, restoration, and alike. We humans have shown that we can mobilize very quickly, in very fundamental ways, when we perceive an existential threat. This is clearly the case with the COVID-19 pandemic. If we can do it for a pandemic, we can do it for existential threats that come from environmental destruction; here I’m thinking mainly about climate change and the biodiversity crisis. There are positive messages even in this horrible situation that we’re experiencing right now.
