
Dr. Karen Lips
Lips was no stranger to chytrid fungus. As a graduate student studying amphibians in Coast Rica and Panama in the 1990s, she witnessed Bd’s devastation first-hand in work that contributed to its discovery. She has since become one of the world’s leading experts in amphibian diseases, and she views the recent discovery of Bsal as a second chance to slow down the spread of a lethal pathogen before it is too late. In a study published in the journal Science the fall of 2014, Lips and a team of international experts tested more than 5,000 amphibians from four continents for Bsal. They also assessed their vulnerability to Bsal. Though they did not find any Bsal among the tested specimens, they determined that it will be lethal to at least a dozen European and North American salamanders. We were thrilled to have a chance to chat with Lips about this and other exciting research that is coming out of her lab.
How do the pathogenic fungi Batrachochytrium dendrobatidis (Bd) and Batrachochytriumsalamandrivorans (Bsal) infect amphibians? What actually happens to an amphibian once it is infected?
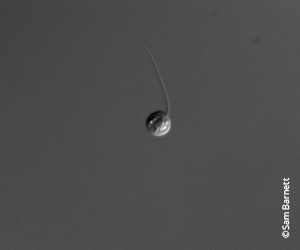
Bd zoospore
Let me preface this by saying that we know much more about Bd and how it works in amphibian skin than we know about Bsal. At this point, we have to assume that the way Bd works in amphibians is how Bsal works in salamanders, because Bsal is so new and there is not much research.
These chytrid fungi have a couple of different stages to their life cycle. The stage that is mobile is the zoospore. It looks like a sperm or tadpole, and it needs water to move around. That’s the stage at which it burrows into the skin of an amphibian.
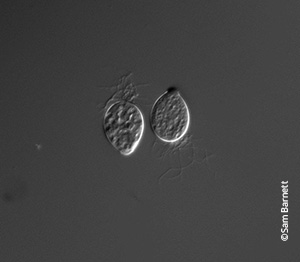
Bd thallium
Once embedded, it goes through metamorphosis into a thallium, which is shaped like a vase. As it matures, it produces more zoospores, which get released back into the environment.
Most of the zoospores fall right back onto the skin of the infected animal and reinfect it. It becomes a race between the growth and rate of reproduction of the fungus and whether or not the amphibian can do anything–behaviorally, chemically, or genetically—to fight it off.
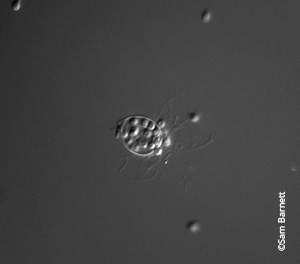
Bd thallus releasing zoospores
There are different strains of the fungus, and those strains grow at different rates.Different temperatures help it grow faster or slower. When it grows fast, the animal is unable to fight it off. The fungus then physically blocks the skin, and the animal cannot get rid of C02 or take up oxygen. The animal’s blood chemistry gets totally out of whack, which affects organ function. Eventually, if affects the heart to the degree that the heart stops working. It’s somewhat of a slow death. With Bd, we also know that it secretes a chemical we have yet to describe, which turns off lymphocyte production. Lymphocytes are your immune system. So Bd (and probably Bsal) creates this compound that turns off the amphibian’s immune system and allows Bd reproduce faster than the animal can fight it.
Can you tell an amphibian is infected with Bd or Bsal just by looking at it?

One Panamanian golden frog (Atelopus zeteki) died on the day of this Leaf Litter interview
We currently have some Bd infected Panamanian golden frogs (Atelopus zeteki) in an experiment. They have been infected [at varying levels] for four weeks, and some are just starting to die. We have watched them every single day. We can tell right before they die that they are really bad because they become extremely lethargic, but until that point, the impact of the disease is not visible.
Does the disease usually kill within four weeks?
There are over 7,000 species of amphibians. There are seven [known] species of chytrid fungi that infect amphibians. Within a particular species, there are genetic variations. Each one grows differently. Some grow super, super fast. The one that we infected the Panamanian frogs with is the worst. That’s what we call the GPL2 (global pandemic lineage) version. We believe it has spread rapidly and quickly all around the world. When it moves in, it infects everybody, and everybody dies. In culture, it grows incredibly fast. So what we currently understand about how quickly Bd (and possibly Bsal) kills is at one end of the continuum.
GPL2
Then we have a lineage of Bd we call GPL1, which is also of global pandemic lineage, but is slightly different genetically. It is found in various places here in North America. What has it done to our amphibians? We don’t really know. We have gone to museums, and determined that this lineage [strain?] has been around for over a hundred years, and was infecting frogs in Illinois in 1888.
Did those frogs survive?
We don’t know. Imagine what was going on in the U.S. in the 1800s. There was massive deforestation and agriculture. Was that chytrid fungus an invasive species in the 1800s that caused massive die-offs? We don’t know. We really don’t know the ancient history of chytrid fungi, in terms of what they have done to amphibians. We’ve really only seen in the past 20-some years what certain (invasive) lineages have done.
We first thought chytrid fungus was a new thing, and it was killing off everything it came into contact with. Now that we’ve been searching, we have found that while that’s true in some areas, other forms have been around for a long, long time in other areas. In those cases, we don’t know what they’ve done, if they were lethal, or if they’ve lost their virulence over time because they’ve co-evolved with amphibians. Chytrid fungus is a much more complicated story today than it was 20 years ago.
Is there any amphibian champion that is immune to Bd?
No. Sadly, more than 520 species have been found to be infected with Bd.
Does climate change have any effect on Bd’s ability to grow and spread?
We know climate influences the growth of the fungus, as well as the growth and survival of frogs. Most amphibians need a moist, cool environment. Well, that is exactly what the fungus likes, too. So places where frogs do best tend to be where the fungus does best, unfortunately. The hotter you get, the less likely Bd is to thrive. If you can slow the growth of a fungus down, you can live with a low-level infection. It’s when the infection is so high and the skin is blocked, that Bd becomes fatal.
Bsal likes things even cooler, about five degrees lower. This makes sense because the animals on which it is found are mostly of temperate distribution.
So does climate change cause disease? We don’t think so, but certainly different climates are more likely to have more disease or worse impacts than other areas.
Are occurrences of Bd and Bsal mapped on line?
A map of global incidence of positive Bd collections can be found on Bd-Maps. You can see where everyone has ever sampled. They have not created a new site for Bsal, but that is coming.
What can you tell us about the difference between Bd and Bsal?
Bsal was only described a year and a half ago, so we know much less about it. The biggest difference that we know of so far is that Bsal only seems to affect salamanders. Bd is a generalist. It affects all sorts of amphibians all over the world. Bsal really seems to be a problem for the family of newts, Family Salamandridae, which are very susceptible to Bsal and most of the newt species tested are very susceptible to infection by Bsal and show high mortality. Those are signs that Bsal is likely to cause high mortality and population declines in wild populations of newts.

Bd infected fire salamander ((Salamandra salamandra) with lesions
Here in the U.S. we have several species of newts, several of which are extremely abundant and probably play important roles in pond, forest and stream ecosystems. The Martel paper (2014) also showed that at least 17 species and three other families of salamanders can be infected with Bsal, although the other 6 families have not been tested. Because one of the susceptible families is a very ancient group, and because Bd can infect so many species, there is a good chance that Bsal will be shown to infect many salamander species. One other difference is that Bsal causes lesions. I do not know if that’s in 100% of animals, but at least in the fire salamanders in Europe, you can see open wounds.
I read that Bsal is believed to have originated in Southeast Asia 30 million years ago, but only recently arrived in Europe. How did it get there?
Bsal has been found in the wild in three places in Asia: Thailand, Japan, and Vietnam. They also found it in museum specimens of Asian newts from over 150 years ago. So we know it’s in the field in Asia, and that it has likely been there for a long time, but we don’t know what it is doing to animals there.
Is Bsal not killing newts in Asia?
There are losses of newts in Asia, but many of these newts are heavily, heavily collected for the pet trade. So the question is: are they not there because somebody scooped them up and sold them, or because they died from Bsal or Bd? Or of something else entirely? There really is no data.
We do know that Bsal is in the pet trade. Animals that were being imported into Europe were swabbed and a couple of those were infected. So that, along with Bsal’s long history in Asia and the huge numbers of animals in the pet trade out of Asia, has been cited as evidence of the movement of Bsal into Europe from Asia.
Right now in Europe, there are two places in Belgium where fire salamanders have the lesions and are dying off. Based on our research, we think that those are the only places in Europe where Bsal is. Hopefully it’s true that Bsal is not here in the U.S. But if it is already all over Asia, and if it is in the pet trade, it is only a matter of time before it gets here. We did that first round of testing in 2014, and now many others are trying to really figure out if it is not here. The good thing is that many people have existing swabs from previous studies. They can go back to those little samples in the freezer and retest them for Bsal. People are also starting new projects in the field and in the lab.
For the 2014 study, you and colleagues tested more than 5,000 amphibians from four continents. How do you actually go about screening a salamander for Bsal?
 It’s actually pretty easy. If you watch CSI, or any of those crime investigation shows, it’s the same thing. We go out in the field wearing gloves, and we catch an animal. We put the animal in a plastic bag, we take something that looks like a Q-tip, and we swab the animal by rubbing the tip over the animal’s skin. Remember, the fungus lives in the skin, and it’s constantly dumping out more zoospores, so when we rub the skin, we’re sort of sucking up those zoospores. It’s just like the CSI team does with blood samples, only we’re looking for Bsal DNA.
It’s actually pretty easy. If you watch CSI, or any of those crime investigation shows, it’s the same thing. We go out in the field wearing gloves, and we catch an animal. We put the animal in a plastic bag, we take something that looks like a Q-tip, and we swab the animal by rubbing the tip over the animal’s skin. Remember, the fungus lives in the skin, and it’s constantly dumping out more zoospores, so when we rub the skin, we’re sort of sucking up those zoospores. It’s just like the CSI team does with blood samples, only we’re looking for Bsal DNA.
The swabs are sent to the lab, and the lab runs the genetics on it. They designed primer which gets mixed with your sample. If your sample contains Bsal, that probe attaches to it, duplicates, and becomes more abundant, and we can say it’s there. If it’s not there, it has nothing to attach to, and it’s a negative.
[For the 2014 study] I sent in all of the North and Central American samples [over 1,400]. We have lots of projects, so getting swabs is not a problem. Up until 2003, when they came up with the genetic tool that allowed us to do swabs, we had to kill the animal and prepare slides from their skin. This took forever and was disruptive to the animals. Since 2003, we have been swabbing hundreds, if not thousands, of animals at our site in Panama. We keep the swabs in a freezer. We did a project in the Appalachians a couple of years ago where we had thousands of swabs of animals. We have also been swabbing museum specimens.
For the 2014 Bsal research, it was like “Let’s move now. Let’s see what’s out there. We can do more, new samples later.” That’s where we are now. We are applying for grants so we can monitor populations and see if they’re declining.
News of your 2014 study made headlines in scientific and mainstream news outlets. To what do you attribute the amount of media attention?
We had a great team of Europeans and Americans, which included [the University of Maryland’s] communications officer at the time, who was an ex-reporter and knew how to promote news like this. But the topic of amphibians and disease had been lurking for a long time, and had become rather high profile.
Bsal represents a “second chance.” When Bd popped up 20 years ago, we had no idea what it was. There was no precedent for having any sort of fungal disease in the skin causing die offs and extinctions. We spent so much time just trying to figure out what was going on. It took years to actually find it in the skin because no one was looking for that. We’ve now had 20 years to study Bd. Bsal is so similar in terms of its biology and what it does. We now have an entire world of people who know how to go in the field and sample, labs designed to instantly run those samples, and people who know how to analyze the data. Everything is in place. We have a second chance to do it right.
In this particular case, the U.S. is way more involved because they can look at Bsal and say, “You’re telling us it’s not here. We can do something about this.” It is very difficult for Fish and Wildlife, or Congress to understand what it is we should do once it is already here. It’s a microbe. You can’t see it, and once it is in the soil and water, we have no way to get rid of it.
I find it very difficult to believe that anything we do is going to be 100% effective, but it might give us enough time to minimize risk, understand how it’s going to move, and really do our best.
If you were in charge of U.S. policy, what would you do to prevent the spread of Bsal?

Panamanian golden frog (Atelopus zeteki)
I’ve spent a lot of time thinking about this, because I had 20 years with Bd. With Bd, we thought lots of things and tried to come up with lots of plans. We can keep frogs alive in captivity but once these fungi get into the environment we have no way of getting rid of them. Maybe some miracle thing will pop up for Bd, but we’ve been looking for 20 years and we’ve found nothing that will work for wild animals. That tells me that anything we can do to keep Bsal out is our most important first action.
Here in the U.S., there is no requirement to test every live, imported animal for disease or pathogens. There are CDC rules for primates, and there are USDA rules for livestock. There are U.S. Fish & Wildlife regulations for certain injurious wildlife, but they fall under the Lacey Act, which was written over 100 years ago, and was designed for things like invasive fish, rats, and crayfish—not microbes. There is hope that Fish & Wildlife can put in a new rule.
Is that in the works?
When the 2014 paper came out, I went down [to Washington, DC] and met with Fish & Wildlife about Bsal. I said, “There’s a new fungus you should be aware of it. It’s not here yet, and it looks like you can do something about it.” They are on record as being concerned. We were able to get a meeting with [USF&W Director] Dan Ashe, and he is concerned. They are studying it and gathering data from a lot of scientists. It sounds like they are moving forward. It is not clear what they will propose at this point.
Congress can do something more generally. There was a bill in both houses last Congress that would require the testing of live animal imports for parasites and pathogens. We think it will be introduced this time around. If passed, that would then give Fish & Wildlife some sort of authority to do something about it. I recently wrote a blog post that includes links to these initiatives.
Speaking of blog posts, I happened to read one powerful post your wrote about your experience discovering Bd in Panama. You basically saw frogs die right before your eyes. Tell us about that.
 I was doing my dissertation in Costa Rica, on the border with Panama. The research site was spectacular. It was a two-hour hike from the house, in the middle of nowhere, at the top of the watershed, with quetzals everywhere. I returned one year, and all the frogs were gone. I found five or six dead animals and collected them, but no one could figure out what happened. [The people in the lab] noticed something in the skin, but they didn’t think that killed them because at that point, Bd had not yet been described.
I was doing my dissertation in Costa Rica, on the border with Panama. The research site was spectacular. It was a two-hour hike from the house, in the middle of nowhere, at the top of the watershed, with quetzals everywhere. I returned one year, and all the frogs were gone. I found five or six dead animals and collected them, but no one could figure out what happened. [The people in the lab] noticed something in the skin, but they didn’t think that killed them because at that point, Bd had not yet been described.
So I switched to a site in western Panama. I was there for a couple of years, and I came back one Christmas and saw 50 dead frogs. That was the smoking gun. This time, we had so many bodies we could send to the pathologist, and we could really describe what we found in the skin. A picture went into the newspaper and people from the National Zoo who had described [a skin infection that was killing frogs] saw it and said, “We have that, too!” That’s how we all got together [in discovery of the chytrid fungus]. So then I moved east to another site in Panama called El Copé. I was there for eight years before Bd got there. Every summer, we’d go down for three months and swab everything. One day, there were dead frogs, and that was the end.
On the one hand it was great in that we predicted the arrival of Bd, and being able to predict it told us that it really was the disease. It enabled us to rule out things like climate change.
 Because I have been working with Bd for so long, I’m so invested in the topic–personally and professionally. All of us who work in this field have very conflicted emotions. On the one hand, as a scientist, it is exciting to discover something. But the fact that we discovered Bd was very bad for the animals we’ve been working with. Every bit of scientific happiness comes with conservation sadness.
Because I have been working with Bd for so long, I’m so invested in the topic–personally and professionally. All of us who work in this field have very conflicted emotions. On the one hand, as a scientist, it is exciting to discover something. But the fact that we discovered Bd was very bad for the animals we’ve been working with. Every bit of scientific happiness comes with conservation sadness.
Are there other pathogens are you worried about, when it comes to salamanders?
Currently, no. But the way things are going with chytrid fungi, I think we need to be prepared for almost anything. We described Bd in 1998. Bsal was described a year and a half ago. In those intervening years, we found six other species of Bd. So that’s eight new pathogens of amphibians in less than 20 years. It makes you wonder how many more are out there.
In addition to studying diseases, your lab studies the impacts of amphibian decline on ecosystems. I know from speaking with Ray Semlistch at the University of Missouri that we still have a lot to learn about the role of salamanders in forest ecosystems. What information can you share with us about the role that salamanders can or may play in certain ecosystems and the impact Bsal might have on ecosystems?

OLYMPUS DIGITAL CAMERA
I have been involved in two projects related to the role of amphibians in ecosystems. One involved [the genus] Ambystoma. Those are pond breeding, mole salamanders like the tiger and spotted salamanders. I had a student who studied a bunch of ponds in Illinois. He had three species, and they all come out of the woods and into the same ponds to breed at different times. What he found is that most of the eggs hatch into larvae, but most of the larvae die and basically feed the pond. So very few of these animals actually leave the pond, go back into the forest and come back the next year. The salamanders are putting energy into the pond. The larvae eat things and move things around, and then they die in the pond and become food. Or, one salamander will eat other salamander larvae. So it turns out salamanders are very important to energy flow in these pond forest ecosystems.
The other project tadpoles in streams in Panama. When Bd arrived, it wiped out all the frogs and tadpoles. We found that when all the tadpoles were removed from those streams, algae grew that was not there before. There was a dead slime layer because there were no tadpoles to eat it. This affected the aquatic invertebrates in the stream, like stoneflies and mayflies, which are food for fish and birds. Everything we measured in that stream changed because of [the arrival of Bd]: water quality, sediment, algae, diatoms, insects, etc.
And that didn’t happen over decades, right?
It happened very quickly. We were lucky that we had, by pure chance, set up this project, and then Bd arrived. We were able to do before/after comparisons. We’ve shown that it affected the nutrient cycling. We think it also affected the other vertebrates, like snakes. Many disappeared. They either starved or shifted their habitat. The impacts were probably even greater than we were able to measure.
We’d like to do some work in the Appalachians. There have been a handful of studies there–some very classic—but we’re just at the tip of the iceberg. If you have ever gone out there and flipped rocks, you know that red-backed salamanders (Plethodon cinereus) alone are, like, under every rock. And you’re probably seeing less than 10 percent of what is out there. Imagine how important they must be to recycling nutrients, and eating all of the little bugs in the leaf litter.
Do you have hope for salamanders?
Maybe we’ll find a cure for these microbial things. The true test is coming up with something that can cure them in the wild. But [my real source of hope is knowing] that evolution happens. The more we learn, the more we see that evolution has already been in the works. Perhaps that’s why, for example, in Illinois, where some form of Bd has been around for 120 years, we’re not seeing die offs. We also know, based on recent lab work, that if you [infect] an animal multiple times, they do better the fourth or fifth time than they did the first time. It’s still fairly early on in the story, but we are beginning to see differences among species and among populations in terms of how they respond to these fungi. We know evolution works, but the question is: can it work fast enough?

For more about the Lips Lab at the University of Maryland, check out this video or visit the Lips Lab web site.

